Anti-IgE Treatment for Severe Allergic Asthma
Advanced Biologic Therapy for Severe Allergic Asthma
Anti-IgE treatment is an advanced biologic therapy used for patients with severe allergic asthma that remains poorly controlled despite high-dose inhaled therapy. This treatment targets the underlying allergic mechanism, rather than only relieving symptoms.
At our clinic, Anti-IgE therapy is prescribed and monitored by experienced allergy and immunology consultants, following a comprehensive clinical assessment.
What Is Anti-IgE Therapy?
Anti-IgE therapy uses a humanised monoclonal antibody that binds to free immunoglobulin E (IgE) in the bloodstream. By neutralising circulating IgE, the treatment:
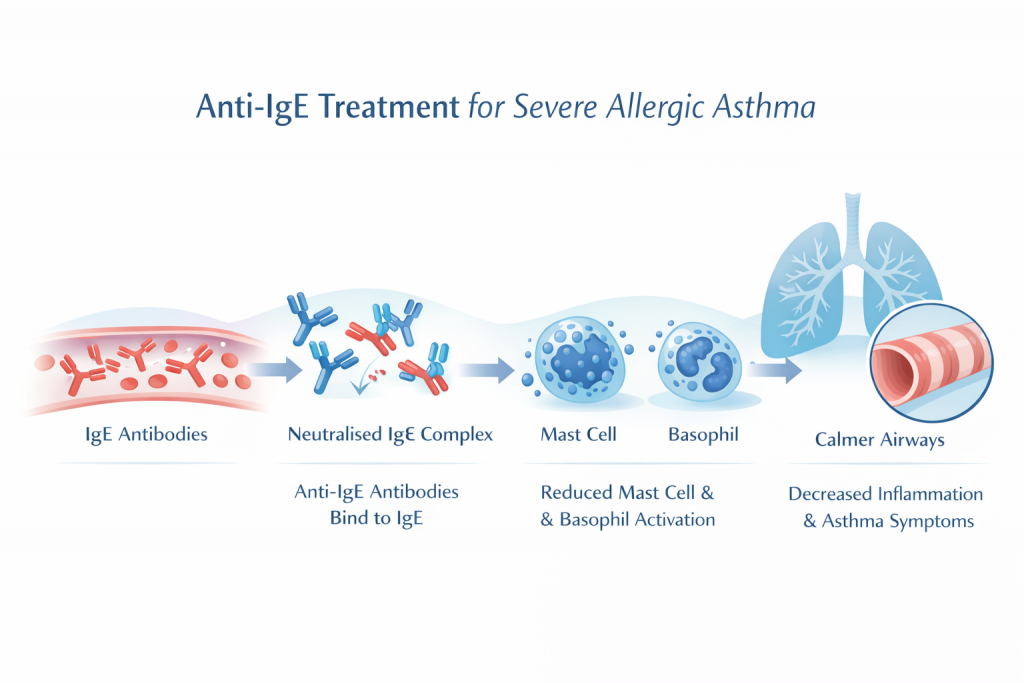
Anti-IgE therapy reduces allergic inflammation and improves asthma control.
– Reduces activation of mast cells and basophils
– Lowers expression of IgE receptors on inflammatory cells
– Decreases allergic airway inflammation
– Reduces asthma exacerbations and hospital admissions
As a result, many patients experience improved asthma control, fewer flare-ups, and a better quality of life.
How Is Anti-IgE Treatment Given?
Anti-IgE therapy is administered as a subcutaneous injection, usually:
– Every 2–4 weeks
– In a clinical setting
– Under specialist supervision
The dose and frequency depend on body weight and baseline IgE levels. Treatment is long-term and requires regular review to assess effectiveness and safety.
Who May Benefit from Anti-IgE Therapy?
Anti-IgE treatment may be suitable for patients who have:
– Severe persistent allergic asthma
– Evidence of IgE-mediated sensitisation
– Frequent asthma exacerbations despite maximal inhaled therapy
– Poor symptom control affecting daily life
There are specific indications and contraindications, which are carefully assessed during your consultation.
Safety, Monitoring and Contraindications
Anti-IgE therapy is generally well tolerated. However:
– Not all asthma is allergic in nature
– Not all patients with allergic asthma are suitable candidates
– Ongoing monitoring is essential
Before starting treatment, patients undergo a full allergy and respiratory assessment, including review of asthma history, previous treatments, and relevant investigations.
Evidence Base and Clinical Effectiveness
Anti-IgE therapy is supported by a strong body of international clinical evidence across multiple allergic conditions, including asthma, allergic rhinitis, and chronic urticaria.
Key peer-reviewed publications include:
– Anti-IgE treatment: a review of its use in the management of allergic asthma
Bang LM, Plosker GL. Treat Respir Med. 2004;3(3):183–199.
– Spotlight on Anti-IgE treatment in allergic asthma
Bang LM, Plosker GL. BioDrugs. 2004;18(6):415–418.
– Anti-IgE for asthma in inner-city children
New England Journal of Medicine. 2011;364:2556–2558.
– Clinical efficacy of Anti-IgE treatment in elderly patients with severe atopic asthma
Allergy and Asthma Proceedings. 2011;32(5).
– Efficacy and safety of Anti-IgE treatment in chronic urticaria
Maurer et al. Journal of Allergy and Clinical Immunology. 2011.
Recent Literature – Anti-IgE Treatment in Severe Allergic Asthma
-
Global Initiative for Asthma (GINA). Global Strategy for Asthma Management and Prevention (2025 update). Published 2025. Accessed February 7, 2026. https://ginasthma.org/wp-content/uploads/2025/11/GINA-2025-Update-25_11_08-WMS.pdf
-
Agache I, Akdis CA, Akdis M, et al. EAACI Biologicals Guidelines—Recommendations for severe asthma. Allergy. 2021;76(1):14-44. doi:10.1111/all.14425. https://pubmed.ncbi.nlm.nih.gov/32484954/
-
Agache I, Rocha C, Beltran J, et al. Efficacy and safety of treatment with biologicals (benralizumab, dupilumab and omalizumab) for severe allergic asthma: A systematic review for the EAACI Guidelines—recommendations on the use of biologicals in severe asthma. Allergy. 2020;75(5):1043-1057. doi:10.1111/all.14235. https://pubmed.ncbi.nlm.nih.gov/32064642/
-
Agache I, Beltran J, Akdis C, et al. Efficacy and safety of treatment with biologicals (benralizumab, dupilumab, mepolizumab, omalizumab and reslizumab) for severe eosinophilic asthma: A systematic review for the EAACI Guidelines—recommendations on the use of biologicals in severe asthma. Allergy. 2020;75(5):1023-1042. doi:10.1111/all.14221. https://pubmed.ncbi.nlm.nih.gov/32034960/
-
Casale TB, Luskin AT, Busse W, et al. Omalizumab effectiveness by biomarker status in patients with asthma: Evidence from PROSPERO, a prospective real-world study. J Allergy Clin Immunol Pract. 2019;7(1):156-164.e1. doi:10.1016/j.jaip.2018.04.043. https://pubmed.ncbi.nlm.nih.gov/29800752/
-
Bousquet J, Humbert M, Gibson PG, et al. Real-world effectiveness of omalizumab in severe allergic asthma: A meta-analysis of observational studies. J Allergy Clin Immunol Pract. 2021;9(7):2702-2714. doi:10.1016/j.jaip.2021.01.011. https://pubmed.ncbi.nlm.nih.gov/33486142/
-
Papaioannou AI, Mplizou M, Porpodis K, et al. Long-term efficacy and safety of omalizumab in patients with allergic asthma: A real-life study. Allergy Asthma Proc. 2021;42(3):235-242. doi:10.2500/aap.2021.42.210014. https://pubmed.ncbi.nlm.nih.gov/33980337/
-
Huang WC, Fu PK, Chan MC, et al. The long-term effectiveness of omalizumab in adult patients with severe allergic asthma: Continuous treatment versus boosting treatment. J Clin Med. 2021;10(4):707. doi:10.3390/jcm10040707. https://pubmed.ncbi.nlm.nih.gov/33670157/
-
Kavanagh JE, Hearn AP, Jackson DJ. A pragmatic guide to choosing biologic therapies in severe asthma. Breathe (Sheff). 2021;17(4):210144. doi:10.1183/20734735.0144-2021. https://pubmed.ncbi.nlm.nih.gov/35296105/
-
Kotoulas SC, Tsiouprou I, Tsaroucha A, et al. Omalizumab: An optimal choice for patients with severe allergic asthma. J Pers Med. 2022;12(2):165. doi:10.3390/jpm12020165. https://pubmed.ncbi.nlm.nih.gov/35207654/
-
Menzella F, Fontana M, Galeone C, et al. Efficacy and safety of omalizumab treatment over a 16-year follow-up: When a clinical trial meets real life. J Asthma Allergy. 2022;15:979-990. https://pmc.ncbi.nlm.nih.gov/articles/PMC9039243/
-
Al Ahmad M, Almohaimeed S, Alenazi A, et al. Real-life effectiveness of omalizumab in patients with severe allergic asthma: (RELIEF study). 2022. https://pubmed.ncbi.nlm.nih.gov/37273950/
Book a Specialist Consultation
Anti-IgE therapy is a prescription-only biologic treatment and must be initiated by a specialist.
Please register as a new patient to arrange a consultant assessment and discuss whether Anti-IgE therapy is suitable for you.
